Initial Design Specifications
Our target is to dilate the cricoid cartilage to be 30% larger than when in its narrowed condition. In its narrowed state, the height of the cricoid region is 3-4 cm. with a diameter of 20-24 millimeters (using males as a reference for now). Thus, with a 30% increase, the stent should push the cartilage to a diameter of 26-30 mm. The stent will dilate for approximately 5-10 minutes, inflating up to 7-8 atm., followed by an ~ 10-minute relaxation. This cycle will repeat for 2-4 weeks. The non-scarred tissue will also be radially pushed outward, leading to some thinning of the tissue. However, it cannot be too thin such that it completely loses its supportive characteristics. Additionally, pressure cannot be put on the vocal cords. The stent must be biocompatible and very hydraulically strong; we are unable specific materials for this feature at the moment. Power for the stent will likely come from Arduino or Raspberry Pi. This power source will be attached externally to the skin and connected to the stent through the tracheostomy tube. Lastly, the stent itself must stay in place in the airway. The shape of the top of the stent must match that of the top of the cricoid cartilage such that it fits snugly. Additionally, the stent may be tied externally through a button-like suture on the neck. Dr. Paniello provided the above numerical metrics and design specifications.
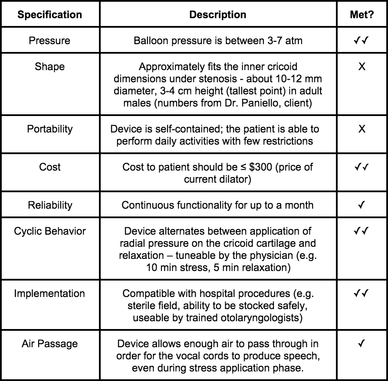
Updated Design Specifications for V&V Testing & Prototype
Two check marks indicates we fully met the specification, one check mark means we met the specification as proof of concept only, and an X indicates we did not meet the specification.
Design Safe
| Design Safe Table.xlsx | |
| File Size: | 38 kb |
| File Type: | xlsx |